In this experiment, three different metals were used: copper, palladium and ruthenium. It is cool to work with the same ligand on different metal centers, as it taught me a lot about what makes them complex in similar manners. It was hard for me to get a good grasp of hard and soft acid base theory, but that improved later on. This was a very fun a simple lab, and it was cool to learn about DMSO.
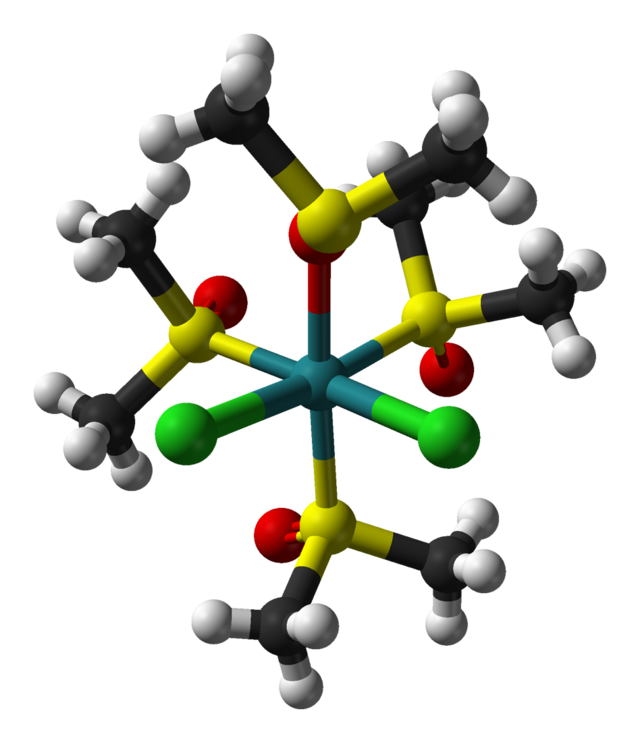
This is the structure of Ruthenium-DMSO complex with four DMSO ligands and two Cl ligands.
Comments
Post a Comment